Getting Attached
In the final part of this 3-part blog series, Defining the undefined with Vitronectin XF™, get attached to Vitronectin even more as we delve into the performance data.
In our previous blogs, we discussed how mixed extracellular matrix (ECM) gels such as Matrigel®, Cultrex®, ECMatrix™, and Geltrex™ are made up of a mix of ECM proteins plus other components. This complex mix, while excellent at supporting a variety of human stem cells in culture, creates a significant headache for potential therapeutics as the exact composition of the gel is not well defined, and there are variations from lot to lot. This lack of definition and inconsistency over time are not easily reconciled with the FDA’s requirements for definition and consistency in any new product. This results at best in delays and costly additional studies prior to starting clinical trials, and at worst rejection of the investigational new drug (IND) application.
Vitronectin XF represents an alternative ECM growth matrix for stem cell-based therapeutics – a xeno-free, single component, defined ECM that satisfies all of the FDA requirements for consistency and reproducibility, avoiding delays and additional work during the regulatory approval process. In this blog, we will see how the performance of Vitronectin XF is equivalent to a complex mixed ECM gel for the culture of human-derived stem cells, promoting cell proliferation, maintaining pluripotency, and supporting normal colony morphology.
The Performance of Vitronectin XF is Equivalent to Matrigel
Matrigel is a commercial product consisting of a gelatinous protein mixture secreted by Engelbreth-Holm-Swarm mouse sarcoma cells and is used as a growth substrate in cell culture. Matrigel consists of collagen IV, laminin, nidogen, and heparan sulfate proteoglycan mixed with other extracellular components and growth factors like TGF-beta and EGF. Because of its heterogeneous composition, it resembles the complex extracellular environment found in many tissues, presenting cultured cells with the adhesive peptide sequences that they would encounter in their natural environment to stimulate complex cell behavior. It is the very complexity of Matrigel that makes it such a good matrix for cell growth.
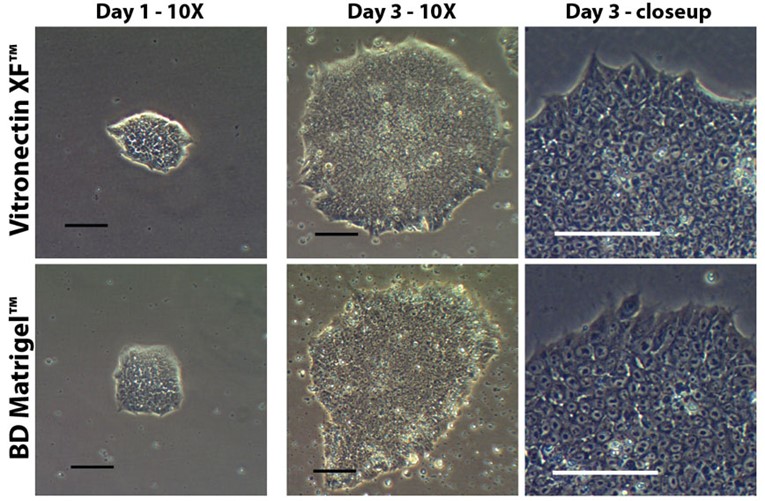
In this set of experiments, we look at the performance of Matrigel against the highly characterized, single component Vitronectin XF and show that despite the complexity of Matrigel, its performance in supporting human induced pluripotent stem cells (hiPSCs) is matched by Vitronectin XF.
Molecular Therapy, Methods and Clinical Development, VOLUME 18, P595-606, SEPTEMBER 11, 2020; Enhancing Chimeric Antigen Receptor T Cell Anti-tumor Function through Advanced Media Design; Saba Ghassemi, Francisco J. Martinez-Becerra, Alyssa M. Master, Sarah A. Richman, David Heo, John Leferovich, Yitao Tu, Juan Carlos García-Cañaveras, Asma Ayari, Yinan Lu, Ai Wang, Joshua D. Rabinowitz, Michael C. Milone, Carl H. June, Roddy S. O’Connor
In this study, cryopreserved hiSPCs were thawed onto BD Matrigel with mTeSR1, and once recovered, were split onto plates coated with either Matrigel or Vitronectin XF. The plates were photographed to track and compare results (see figure 1). Additionally, after 5 passages, the cells were subjected to flow cytometry testing for the pluripotency markers Oct4, SSEA4, and Tra-1-81. In all cases, the cells grown on Vitronectin XF and the cells on Matrigel were shown to be equivalent, demonstrating the ability of Vitronectin XF to support human-derived stem cells at an equivalent level to Matrigel, but without any of the regulatory drawbacks of using a complex mixed ECM gel.
More information on Vitronectin XF and the full data on this comparative study can be found on the Nucleus Biologics website. And in case you missed them, here are part 1 and part 2 in the Defining the Undefined with Vitronectin XF blog series.