The key to every successful therapy is consistency.
Cell therapy manufacturers need solutions that allow them to efficiently harness the ability of a living cell to respond to its environment, while simultaneously delivering replicable and consistent quality performance.
Welcome to our 2-part blog series highlighting Physiologix™, our cGMP, xeno-free media supplement that replaces FBS or human serum. Physiologix helps manufacturers meet their product goals by delivering optimal cellular performance with consistent results.
Cell culture growth media can result in product variability
Cellular therapies are based on living cells, and therefore inherently encompass some level of variability. In some ways, cellular variability is a good thing; the amazing functional plasticity of different cell types allows scientists to find multiple pathways toward achieving a goal, and it allows for flexibility in dealing with the multiple ways in which disease affects each unique individual.
Variability in a therapeutic product, on the other hand, is definitely not a good thing. Treatments need to be consistently effective every time for every patient. In the wake of developmental delays related to several promising new cell therapies [1], the FDA has been urging cell and gene therapy developers to find ways of more diligently characterizing cellular products throughout their lifecycle in order to limit the risk of variable results. [2]
Reproducibility can be partially controlled through stringent quality oversight of methods and instrumentation but controlling variability in cellular starting materials is more difficult. Raw material selection can directly affect the variability, comparability, scalability, and quality of the therapeutic development process – ultimately affecting the cost of therapeutic development and the time it takes to bring these therapies to the market. Even when cells and growth media are meticulously characterized, scale up can change growth parameters and influence cellular performance.
Cell culture media has a profound effect on how cells perform, directly impacting the efficacy of the final cellular product. Nevertheless, cell culture media formulations are often not fully transparent, making it difficult to replicate media in-house, or re-source media should supply https://nucleusbiologics.com/resources/maximizing-quality-and-potency-of-cell-culture-media/line problems arise.
Serum, one of the most commonly utilized protein components in cell culture media, provides all of the growth factors, hormones, cytokines, minerals, and lipids that cells need to thrive and replicate. However, serum is also highly variable, and in fact differs in composition to what cells are exposed to in the body. [3] Not only does it contain thousands of different proteins from multiple cell and tissue types, but the concentration of these components can differ substantially from lot to lot. Understanding the exact composition of media supplements helps to ensure a consistent product and reproducible results.
Physiologix provides validated reliability
Physiologix is a completely xeno-free concentrated growth factor extract made for stem cells and T cells that acts as a replacement for fetal bovine serum or human serum. Physiologix is manufactured under a proprietary GMP manufacturing process with rigorous quality controls that ensure reproducibility of the product. It is extracted from transfusion-grade blood products and comes with a full certificate of analysis, so that composition is transparent to the researcher.
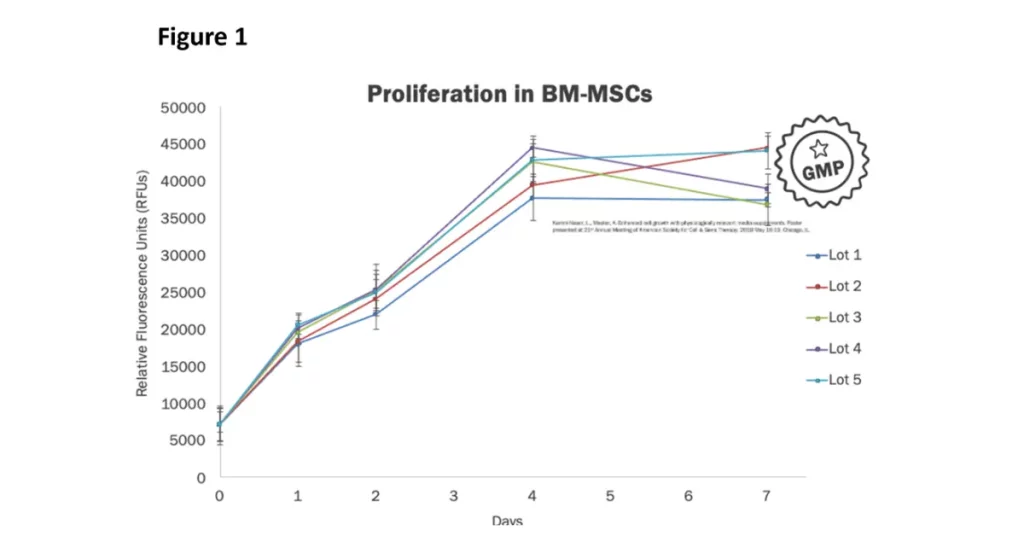
Every lot that is manufactured is delivered with specific quantitative and functional performance characterization data.
In Figure 1, quality control data for bone marrow-derived mesenchymal stem cells (BM-MSC) grown using Physiologix as a serum replacement supplement demonstrates consistent product performance and the ability to efficiently support stem cell proliferation. Each product lot also undergoes sterility testing, adventitious agents screening, and functional proficiency assessment.
Knowing exactly what is in your cell culture media provides peace of mind, but more importantly, a well-characterized product lowers the risk of starting material variability and helps create a more reliable final product. Finally, since our xeno-free media supplement is deliberately formulated to optimize therapeutic cell function [4], researchers can rest assured that they are giving their cell and gene therapy product the best chance of success.
Nucleus Biologics is committed to providing top quality traceable and reproducible cell culture products and services to support cell and gene therapy. Please visit our next blog for Part 2 of our series, where we discuss how using Physiologix helps solve common regulatory compliance headaches, facilitating the transition to commercialization.
References
- Gardner J. FDA gene therapy holdups suggest closer scrutiny by agency. BiopharmaDive, (2020). https://www.biopharmadive.com/news/fda-gene-therapy-manufacturing-delays-scrutiny/588382/
- Pagliarulo N. FDA seeking more consistency from cell, gene therapy developers, top official says. BiopharmaDive, (2021). https://www.biopharmadive.com/news/fda-marks-gene-therapy-consistency/600445/
- Baker, M. Reproducibility: Respect your cells! Nature 537, 433–435 (2016). https://doi.org/10.1038/537433a
- Ghassemi, S., Martinez-Becerra, F. J., Master, A. M., Richman, S. A., Heo, D., Leferovich, J., Tu, Y., García-Cañaveras, J. C., Ayari, A., Lu, Y., Wang, A., Rabinowitz, J. D., Milone, M. C., June, C. H., & O’Connor, R. S. (2020). Enhancing Chimeric Antigen Receptor T Cell Anti-tumor Function through Advanced Media Design. Molecular therapy. Methods & clinical development, 18, 595–606. https://doi.org/10.1016/j.omtm.2020.07.008