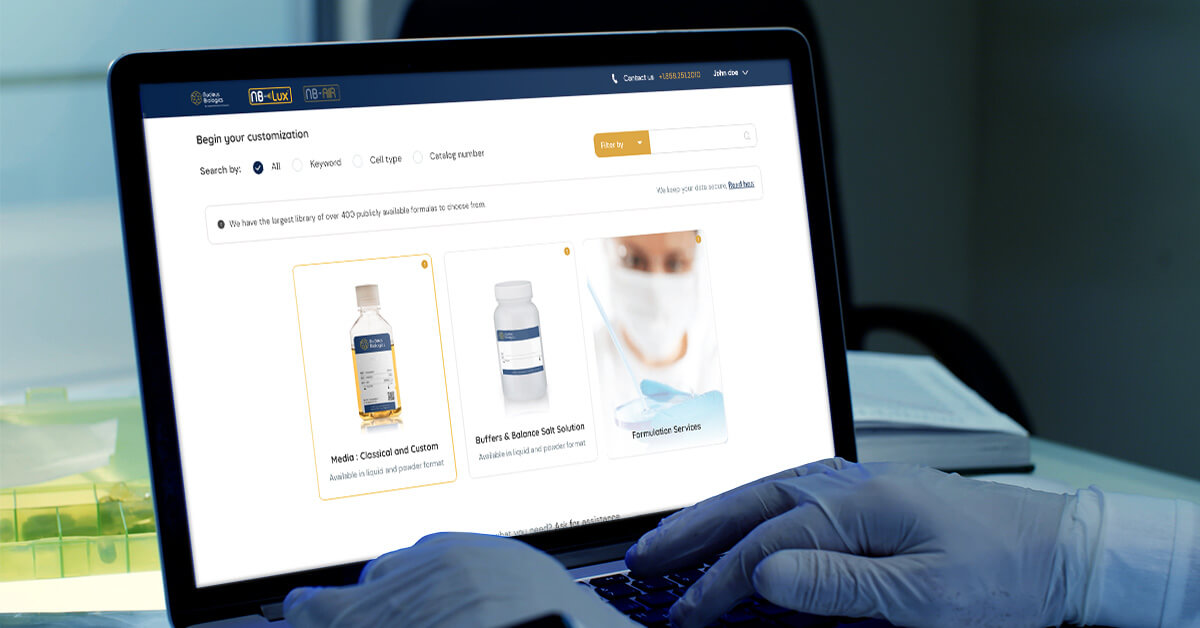
Nucleus Biologics, a leader in precision cell culture, today announced the availability of NB-Lux™, an online configuration and ordering platform for cell culture media. Designed for developers of cell therapies, NB-Lux™ speeds formulation development by providing real time pricing for cGMP grade media in lot sizes from two liters to 2000 liters.
Media is a critical component in the production of cell therapies. During development, companies iteratively test multiple formulations to identify an optimal medium for cGMP manufacturing. Some therapy developers utilize proprietary, off-the-shelf formulations for perceived streamlining in regulatory processes. However, unknown formulations are difficult to tailor to specific therapies, are not part of a therapy company’s intellectual property portfolio and are vulnerable to supply chain issues due to sole sourcing. Equally undesirable, custom media currently takes up to six months to manufacture after it has been formulated.
“Our mission is to speed the time from discovery to cure. We listened to the frustration of our cell therapy customers and reimagined the cell culture media configuration and ordering experience. No longer must a scientist guess what to add to proprietary, sole sourced medias or wait almost half a year to receive custom media,” said David Sheehan, Founder, President and CEO of Nucleus Biologics. “NB-Lux™, a first-of-its-kind portal, empowers a scientist to create their own formulas, get real time quotes, and, in some situations, have cGMP media manufactured in as little as 9 weeks. This is the first step in our plan to reshape the media market. My sense is the COVID-19 pandemic has punctuated the need for the scientific community and industry to create new paradigms that expedite delivery of proven therapies.”
Nucleus Biologics developed the patent-pending NB-Lux™ to provide real time quotes for cGMP cell culture media. Users can input and manipulate different formulations online, receive real time pricing, and track order progress. NB-Lux™ is powered by one of the largest repositories of over 400 publicly available formulations. Users can optimize each individual chemical component, testing requirements, and packaging. By customizing a media to the unique critical attributes of each therapy, developers can take control of formulations, rapidly progress from small- to large-scale production, and move therapies to the clinic faster.
Media Contact:
Alyssa Master
[email protected], (858) 251-2010
About Nucleus Biologics
Nucleus Biologics is a San Diego based biotechnology company and a global provider of media and supplements for cell and gene therapy. Their mission is to speed the time from scientific discovery to cure by driving reproducibility through Precision Cell Culture products and tools. Nucleus Biologics delivers innovative, transparent and cGMP products and services with the goal of disrupting the market and eliminating antiquated practices and products. Ultimately, Nucleus Biologics strives to create a new paradigm that serves both scientists and clinicians, while reducing the environmental footprint of cell culture. www.nucleusbiologics.com